 The last exclusive feature that available on the Professional edition only is its clone HDD or SSD function, where it clones an entire hard drive either to another internal drive or an external one. The modern periodic table is based on Dmitri Mendeleev’s 1896 observations that chemical elements can be grouped according to common chemical properties. Another exclusive feature is the monitor hard drive health function, which allows users to check on their hard drive’s condition in order to avoid any issues with it later on down the line. This feature is useful for people who want to make sure their CDs or DVDs are not corrupted and can be used again. One of the exclusive features is the ability to recover data from CD/DVD. The periodic table is an arrangement of the elements in order of increasing atomic number. With Stellar Data Recovery Professional, you will also get exclusive features that are not available in the standard edition. The software also offers disk imaging tools so you can create image files of your hard drive to protect against future disasters. Stellar Data Recovery Pro also recovers emails that have been deleted to find all of your emails on any drive and return them back intact, so you don’t lose anything important!. The protons and neutrons account for almost all of the mass of an atom. the number of protons in the nucleus of an atom, which determines the chemical properties of an element and its place in the periodic table. With this software, you can recover permanently deleted data, recover data from formatted drives, and recover lost partition recovery. A period 1 element is one of the chemical elements in the first row (or period) of the periodic table of the chemical elements. The group 16 elements each add two electrons, while the group 17 elements add one electron per atom, to form the anions.Stellar Data Recovery Professional is a software that specializes in recovering data from all sorts of media. Notice that the group 16 ions are larger than the group 17 ions. The electron cloud also spreads out because more electrons results in greater electron-electron repulsion. They are organized in a tabular format, where a row represents a period, and a column represents a group. All elements in a row have the same number of electron shells. The periodic table is an arrangement of all known elements in order of increasing atomic number and recurring chemical properties. When the electrons outnumber the protons, the overall attractive force that the protons have for the electrons is decreased. A period on the periodic table is a row of chemical elements. The addition of electrons always results in an anion that is larger than the parent atom. The potassium atom has one electron removed to form the corresponding ion, while calcium loses two electrons. One other factor is the number of electrons removed. 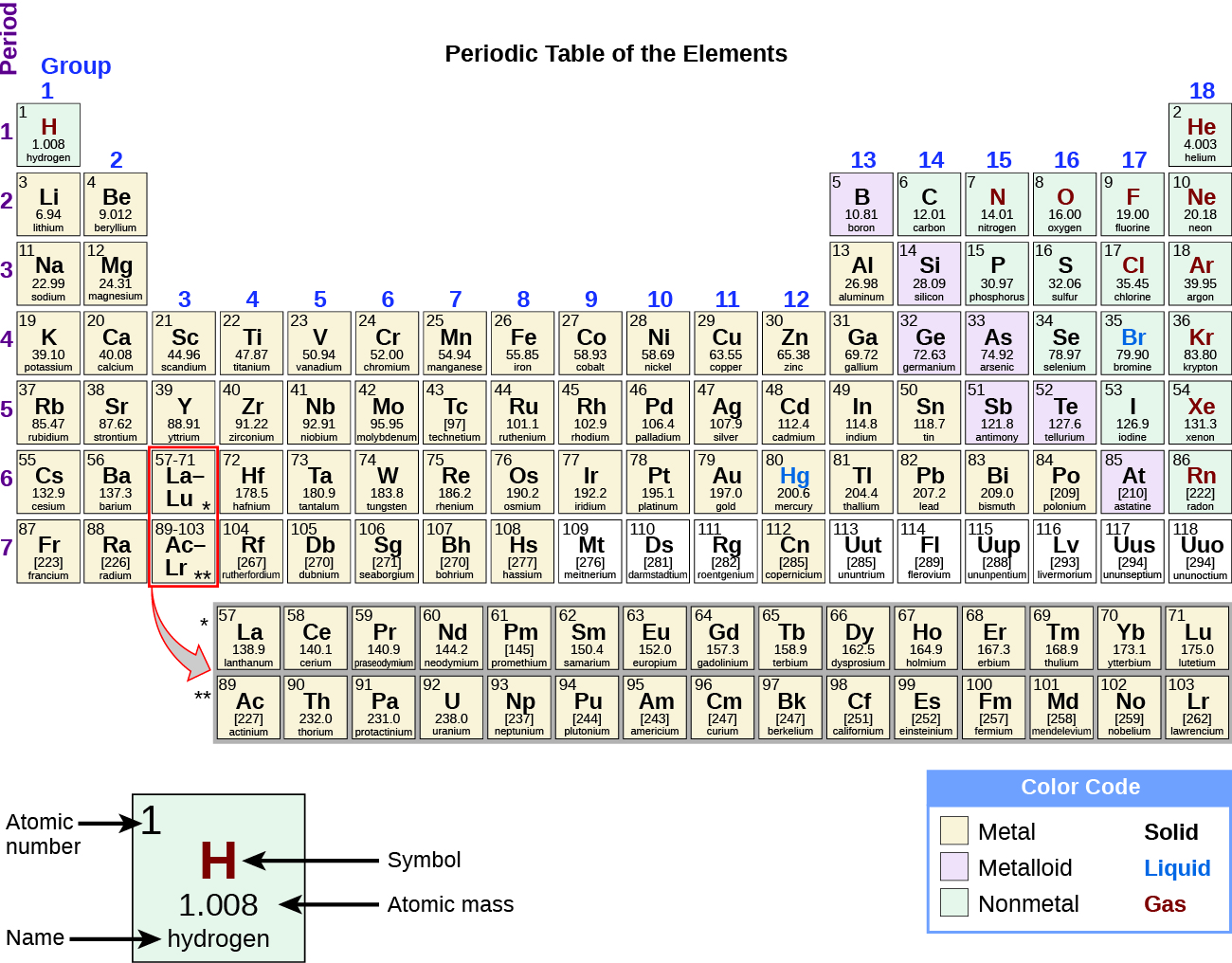
Another reason is that the remaining electrons are drawn closer to the nucleus because the protons now outnumber the electrons. Mendeleev organized elements according to recurring properties to make a periodic table of elements. In each period, the elements atomic numbers increase from. 
Periodicity is caused by regular and predictable variations in element atomic structure. The periodic table contains seven periods 5 (nine if you count the lanthanides and actinide series). When the valence electron(s) are removed, the resulting ion has one fewer occupied principal energy level, so the electron cloud that remains is smaller. In the context of chemistry and the periodic table, periodicity refers to trends or recurring variations in element properties with increasing atomic number. The removal of electrons always results in a cation that is considerably smaller than the parent atom. /periodic-table-of-the-elements-2017--illustration-769723031-5aa02f9b04d1cf00386ccf7c.jpg)
(Credit: Zachary Wilson Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window)) Groups 16 and 17 are nonmetals and form anions, shown in purple. Groups 1, 2, and 13 are metals and form cations, shown in green. The periodic table, also known as the periodic table of the elements, arranges the chemical elements into rows ('periods') and columns ('groups'). \): Comparison of ion sizes for Groups 1, 2, 13, 16, and 17. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
